Water
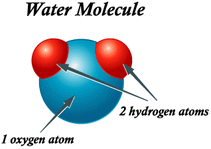
Water is a polar, covalent, inorganic molecule. Polar covalent molecules are covalently bonded particles, in which the electrons are unequally shared between atoms, therefore creating dipoles. Water is inorganic because it does not contain carbon. When the 2 hydrogen atoms, and 1 oxygen atom covalently bond to form H2O, the electrons are unequally shared between the oxygen atom and the hydrogen atoms, due to the greater electronegativity of oxygen. In result. the electrons tend to spend more time with the oxygen atoms, in comparison to the hydrogen atoms, causing the oxygen atom to become slightly negative and the hydrogen atoms to become slightly positive. All these features of water are what make it so important for life on earth. Here are some of its amazing abilities:
High Heat Capacity8. Water is stable between 0°C and 100°C
1. Water has a high heat capacity- |
Water is Neutral |
Transparency |
Ice Floats on Water |
Adhesion
Cohesion
Excellent Solvent
Acids

Bases

PH

Buffers

